|
(For an outline, click here) Interspecific Competition Mechanisms of Competition  World's smallest monitor lizard, a neonate
World's smallest monitor lizard, a neonate Varanus brevicauda , approximately actual size, (SVL = 52 mm, weight 1.4 gms) ---> Competition occurs indirectly when two or more organismic units use the same resources and when those resources are in short supply. Such a process involving resource depression or depletion has been labeled consumptive or exploitative competition. Competition may also occur through more direct interactions such as in interspecific territoriality (interference competition). Competition for space, as in the rocky intertidal, often mediated by who arrives first, has been termed pre-emptive competition. Interaction between the two organismic units reduces the fitness and/or equilibrium population density of each. This can occur in several ways. By requiring that an organismic unit expend some of its time and/or matter and/or energy on competition or the avoidance of competition, a competitor may effectively reduce the amounts left for maintenance and reproduction. By using up or occupying some of a scarce resource, competitors directly reduce the amount available to other organismic units. Mechanisms of interference competition, such as interspecific territoriality, will be favored by natural selection only when there is a potential for overlap in the use of limited resources at the outset (i.e., when exploitation competition is potentially possible). Interspecific competition between individuals belonging to different species is of great interest. Because of its symmetry, it is always advantageous, when possible, for either party in a competitive relation to avoid the interaction: competition has therefore been an important evolutionary force that has led to niche separation, specialization, and diversification. If, however, avoidance of a competitive interaction is impossible, natural selection may sometimes favor convergence. Competition is not an on-off process; rather, its level presumably varies continuously as the ratio of demand over supply changes. There is little, if any, competition in an ecological vacuum, whereas competition is keen in a fully saturated environment. All degrees of intermediates exist. In real populations, rates of increase, competitive abilities, and carrying capacities vary from individual to individual, with population density, community composition, and in space and time. Indeed, temporal variation in the environment may often allow coexistence by continually altering the competitive abilities of populations inhabiting it. Time lags are doubtless of some importance in real populations. Finally, a heterogeneous environment may allow real competitors to evolve divergent resource utilization patterns and to reduce interspecific competitive inhibition.  Competitive Exclusion How does one population drive another to extinction? When one species ultimately eliminates the other entirely when the two come into competition and the system is allowed to go to saturation, we say then that competitive exclusion has occurred. Consider an ecological vacuum inoculated with small numbers of each species. At first, both populations grow nearly exponentially at rates determined by their respective instantaneous maximal rates of increase. As the ecological vacuum is filled, the actual rates of increase become progressively smaller and smaller. Both populations are infinitely unlikely to have exactly the same rates of increase, competitive abilities, and carrying capacities. Hence, as the ecological vacuum is filled, a time must come when one population's actual rate of increase drops to zero while the other's rate of increase is still positive. This situation represents a turning point in competition, for the second population now increases still further and its competitive inhibition of the first is further intensified, reducing the actual rate of increase of the first population to a negative value. The first population is now declining while the second is still increasing; barring changes in competitive parameters, competitive exclusion (extinction of the first population) is only a matter of time. This process has been demonstrated experimentally Some rather strong statements concerning competitive exclusion have been made. Among them is a hypothesis called the competitive exclusion "principle"; two species with identical ecologies cannot live together in the same place at the same time. Ultimately one must edge out the other; complete ecological overlap is impossible. The corollary is that if two species coexist, there must be ecological differences between them. Since any two organismic units are infinitely unlikely to be exactly identical, mere observation of ecological differences between species does not constitute "verification" of the hypothesis. Untestable hypotheses like this one are of little scientific utility and are gradually forgotten by the scientific community. However, the competitive exclusion "principle" has served a useful purpose by emphasizing that some ecological difference may be necessary for coexistence in competitive communities in saturated environments. Ecologists have gone on to ask more penetrating and dynamic questions: How much ecological overlap can two species tolerate and still coexist? How does this maximal tolerable overlap vary as the ratio of demand to supply changes? How high must migration rates be for competitively inferior fugitive species to persist in spatially and temporally varying patchy habitats? Can temporally changing competitive abilities lead to coexistence? Evolutionary Consequences of Competition Natural selection in saturated environments (K selection) favors competitive ability. A raft of presumed populational products of intraspecific competition over evolutionary time include: rectangular survivorship, delayed reproduction, decreased clutch size, increased size of offspring, parental care, mating systems, dispersed spacing systems, and territoriality. Perhaps the most far-reaching evolutionary effect of interspecific competition is ecological diversification, also termed niche separation. This in turn has made possible, and has led to, development of complex biological communities. Another presumed result of both intraspecific and interspecific competition is increased efficiency of utilization of resources in short supply.  Laboratory Experiments Competition is often fairly easily studied by direct experiment, and many such studies have been made. Gause (1934) was one of the first to investigate competition in the laboratory; his classic early experiments on protozoa verified competitive exclusion. He grew cultures of two species of Paramecium in isolation and in mixed cultures, under carefully controlled conditions and nearly constant food supply. "Carrying capacities" and respective rates of population growth of each species when grown separately and in competition were then calculated (competition coefficients are also readily computed from such data). Interestingly enough, the protozoan with the highest maximal instantaneous rate of increase per individual (P. caudatum) was the inferior competitor, as expected from considerations of r and K selection. An effect of environment on the outcome of competition was demonstrated by Park and his many colleagues. Working with two species of flour beetles (Tribolium), these investigators showed that, depending on conditions of temperature and humidity, either species could eliminate the other. The outcome of competition between two beetle species can be reversed by a protozoan parasite. In early experiments, the outcome of competition under particular environmental conditions could not always be predicted in a set of environments termed the "indeterminate zone." More recently, however, this zone has been substantially reduced by taking into account the reproductive values and genotypes of beetles. Under some environmental conditions, cultures that had begun with a numerical preponderance of a given species always resulted in the extermination of the other species. In a laboratory study of competitive interactions among ciliate protozoans, Vandermeer (1969) cultured each of four species separately and in all possible pairs. These experiments allowed estimation of r's, K's, and alphas. Observed pairwise interactive effects were similar to those actually observed when all four species were grown together in mixed culture -- an indication that higher-order interactions among these species were slight. However, several similar studies (Hairston et al., 1968; Wilbur, 1972; Neill, 1974) suggest strong interactive effects among species, with the competitive effects between any two species depending strongly on the presence or absence of a third. Competition and competitive exclusion have now been demonstrated in laboratory experiments on a wide variety of plants and animals. Potential flaws in many of these investigations are that, for practical reasons, they are carried out in constant and simple environments, almost invariably on small, often relatively r-selected, organisms that may not encounter high levels of competition regularly under natural circumstances. Circumstantial Evidence from Nature Competition is notoriously difficult to demonstrate in natural communities, but a variety of observations and studies suggest that it does indeed occur regularly in nature and that it has been important in molding the ecologies of many species of plants and animals. Even if competition did not occur on a day-to-day basis, it could nevertheless still be a significant force; active avoidance of interspecific competition in itself implies that competition has occurred sometime in the past and that the species concerned have adapted to one another's presence. Also it might be difficult to find competition actually occurring in nature because inefficient competitors should be eliminated by competitive exclusion and therefore might not normally be observable. We might not expect to find abundant evidence of competition in small, short-lived organisms, such as insects and annual plants, but would look for it in larger, longer-lived organisms, such as vertebrates and perennial plants. Ecologists have several different sorts of evidence, much of which is circumstantial, suggesting that competition either has occurred or is occurring in natural populations. These include: (1) studies on the ecologies of closely related species living in the same area; (2) character displacement; (3) studies on "incomplete" floras and faunas and associated changes in niches, or "niche shifts"; and (4) taxonomic composition of communities. Closely related species, especially those in the same genus, or "congeneric" species, are often quite similar morphologically, physiologically, behaviorally, and ecologically. As a result, competition is intense between pairs of such species that live in the same area, known as sympatric congeners; selection may be strong to render their ecologies more different or to lead to ecological separation. Many groups of closely-related sympatric species have been studied, and almost without exception, detailed investigations on relatively K-selected organisms have revealed subtle but important ecological differences between such species. Usually the differences are of one or more of three basic types: (1) the species exploit different habitats or microhabitats (differential spatial utilization of the environment); (2) they eat different foods; or (3) they are active at different times (differential patterns of temporal activity). Such ecological differences are known as "niche dimensions" because they are important in defining a species' role in its community and its interactions with other species. Many examples of clear-cut habitat and microhabitat differences could be cited. MacArthur (1958) studied spatial utilization patterns in five species of sympatric warblers (genus Dendroica) by noting the time spent in precise locations by foraging individuals of each species. Each species has its own unique pattern of exploiting the forest (Figure 12.9). Overdispersion in general, and territoriality in particular, are indicative of competition in that they reduce its intensity. Overdispersion is widespread in plants, as is territoriality in vertebrate populations. Indeed, interspecific territoriality has been documented in many species of iguanian lizards and birds and probably occurs in other taxa as well; thus, both intraspecific and interspecific competition have led to territorial behavior. Differences in time of activity among ecologically similar animals can effectively reduce competition, provided that resources differ at different times. This is true in situations where resources are rapidly renewed, because the resources available at any one instant are relatively unaffected by what has happened at previous times. Perhaps the most obvious type of temporal separation is that between day and night; animals active during the daytime are "diurnal," those active at night are "nocturnal." Examples of pairs apparently separated by such temporal differences are hawks and owls, swallows and bats, or grasshoppers and crickets. Patterns of activity within the course of the day alone also differ, with some species being active early in the morning, others at midday, and so on. Seasonal separation of activity also occurs among some animals, such as certain lizards. In many animals, daily time of activity changes seasonally (Figure 12.11). Dietary separation among closely-related animal species has been shown repeatedly. For example, Table 12.3 shows that several sympatric congeneric species of the marine snail genus Conus (commonly called "cone shells") eat distinctly different foods (Kohn, 1959). Similarly, three species of stoneflies eat prey of different sizes (Figure 12.12). Among desert lizards, diets of several sympatric species are composed predominantly of ants, termites, other lizards, and plants (Pianka, 1966b, 1986). Similar cases of food differences among related sympatric species are known in many birds and mammals. Simultaneous differences in the use of space, time, and food have also been documented for some sympatric congeneric species. In lizards of the genus Ctenotus (Pianka, 1969), for instance, seven sympatric species forage at different times, in different microhabitats, and/or on different foods. Frequently, in such cases, pairs of species with high overlap in one niche dimension have low overlap along another, presumably reducing competition between them. The phenomenon of "character displacement," which refers to increased differences between species where they occur together, is also evidence that competition occurs in nature. Sometimes two widely-ranging species are ecologically more similar in the parts of their ranges where each occurs alone without its competitor (i.e., an allopatry) than they are where both occur together (in sympatry). This sort of ecological divergence can take the form of morphological, behavioral, and/or physiological differences. One way in which character displacement occurs is in the size of the food-gathering or "trophic" apparatus, such as mouthparts, beaks, or jaws. Prey size is usually strongly correlated with the size of an animal's beak or jaw as well as with its structure (Figure 12.13). Although the evidence is circumstantial (see Grant, 1972, for a review), character displacement in either body size or the size of the trophic apparatus is thought to have occurred in some lizards, snails (Figure 12.14), birds, mammals, and insects, presumably separating food niches. Such niche shifts in the presence of a potential competitor suggest that each population has adapted to the other by evolving a means to reduce interspecific competition. Morphological character displacement in the size of mouthparts need not evolve if the populations concerned have diverged in other ways; hence, it is expected only in situations where both competitors occur side by side, exploiting identical microhabitats (i.e., true syntopy). Animals that forage in different microhabitats, such as the warblers of Figure 12.9, have adapted to one another primarily by means of behavioral, rather than morphological, character displacement. Size differences between closely-related sympatric species have been implicated as being necessary for coexistence (Hutchinson 1959), and even in the "assembly" of communities (Case et al. 1983), although there has been considerable dispute over the statistical validity of these patterns (Grant 1972; Horn and May 1977; Grant and Abbott 1980). There may be a definite limit on how similar two competitors can be and still avoid competitive exclusion; character displacement in average mouthpart sizes is often about 1.3, and the length ratio of 1.3 has been suggested as a crude estimate of just how different two species must be to coexist syntopically. (For body mass, a ratio of 2 corresponds to the length ratio of 1.3.). O Among seven species of Australian varanid lizards (Pianka 1994), only two of 21 such Hutchinsonian ratios of head lengths are below 1.3, caudolineatus/brevicauda and eremius/gilleni. Both ratios are between pygmy monitors, and, interestingly both low ratios are between an arboreal and a terrestrial species. V. caudolineatus and brevicauda are not sympatric. Only one gilleni was collected at Red Sands, so this arboreal species may not maintain a viable population, but might merely represent an "accidental" migrant wandering through sandy spinifex habitat from nearby mulga habitat. Perhaps the most thorough analysis of such "Hutchinsonian" ratios is that provided for the world's bird-eating hawks by Schoener (1984), who computed size ratios among all possible pairs and triplets of 47 species of accipiter hawks. Frequency distributions of expected size ratios were generated for all possible combinations of species, which were then compared with the much smaller number of existing accipiter assemblages. Schoener found a distinct paucity of low size ratios among real assemblages, strongly suggesting size assortment. Following Schoener's rationale and methodology, head lengths of 24 species of Australian Varanus were estimated from data compiled on maximum snout-vent length by Greer (1989), as follows. Among the seven desert species (Table 1), which include size extremes as well as members of both major lineages (subgenera) of Australian Varanus, average adult head length is almost perfectly correlated with average adult SVL (r = .999, P<.001). Head lengths of the 24 species were estimated by simple linear regression. 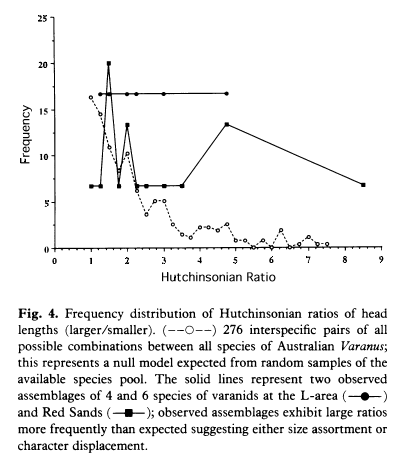
Another type of evidence for competition comes from studies on so-called "incomplete" biotas, such as islands, where all of the usual species are not present. Those species that invade such areas often expand their niches and exploit new habitats and resources that are normally exploited by other species on areas with more complete faunas. On the island of Bermuda, for example, considerably fewer species of land birds occur than on the mainland, with the three most abundant being the cardinal, catbird, and white-eyed vireo. Crowell (1962) found that, compared with the mainland, these three species are much more abundant on Bermuda and that they occur in a wider range of habitats. In addition, all three have somewhat different feeding habits on the island, and one species at least (the vireo) employs a greater variety of foraging techniques. Mountaintops represent islands on the terrestrial landscape just as surely as Bermuda is an island in the ocean, and they often show similar phenomena. For example, two congeneric species of salamanders, Plethodon jordani and P. glutinosus, occur in sympatry on mountains in the eastern United States. In sympatry, the two species are altitudinally separated, with glutinosus occurring at lower elevations than jordani; vertical overlap between the two species never exceeded 70 meters (Hairston, 1951). On mountaintops where jordani occurs, glutinosus is restricted to lower elevations, whereas on adjacent mountains that lack jordani, glutinosus is found at higher elevations, often right up to the peak. Niche expansion under reduced interspecific competition has been termed "ecological release." Further evidence of competition stems from a corollary of ecological release; when mainland forms are introduced on to islands, native species are frequently driven to extinction, presumably via competitive exclusion. Thus, many birds that once occurred only on Hawaii became extinct shortly after the introduction of mainland birds such as the English sparrow and the starling (Pimm and Moulton, 1966). Similar extinctions have apparently occurred in the Australian marsupial fauna (e.g., the Tasmanian wolf, but also many species of mid-sized marsupials) with the introduction of placental mammal species (e.g., the dingo dog and the European fox). Of course, fossil history is replete with cases of natural invasions and subsequent extinctions. The simplest and most plausible explanation for many of these observations is that surviving species were superior competitors and that niche overlap was too great for coexistence. Before natural selection could produce character displacement and niche separation, one species had become extinct. Elton (1958) discusses many other examples of ecological invasions among both plants and animals. Other Prospects Although competition is presumably central to numerous ecological processes and phenomena, current understanding of competitive interactions remains inadequate from both theoretical and empirical points of view. Possibilities abound for significant work. Clearly, the great temporal and spatial heterogeneity of the real world demands a dynamic approach to competitive interactions. Models that depart from the notion of competitive communities at equilibrium with their resources are of interest. The notion of competition coefficients itself may be somewhat illusory and may often obscure the real mechanisms and dynamics of competition. Even so, theory could be improved substantially simply by treating alphas as variables in both ecological and evolutionary time. For example, the actual shapes of resource utilization curves might be allowed to change (subject to an appropriate constraint such as holding the area under the curves constant), either in ecological time by behavioral release or in evolutionary time via directional selection favoring deviant phenotypes. Because the competitive effects between any given pair of species are often sensitive to the presence or absence of a third species, theory is needed on interactive competition coefficients. An attractive alternative to competition coefficients is to measure the intensity of interactions between species by the sensitivity of each species' own density to changes in the density of the other. As such, these interactions are represented mathematically by partial derivatives; if the presence of species j is detrimental to species i, delta N(sub i) over delta N(sub j) is negative, whereas a beneficial interaction has a positive sign. Note that this approach involves population dynamics and that it can be applied to prey-predator and symbiotic interactions as well as to competitive ones. Great potential also exists for further development of theory on diffuse competition. Consider two communities with similar numbers of species but different guild structures. In the first, several distinct clusters of competing species have strong competitive interactions among themselves but weak interactions with members of other guilds. In the second community, all members interact more or less equally and diffuse competition is more intense. (Differences in the degree of niche dimensionality would produce such a difference between communities. What effects will such differences in degree of competitive "connectedness" have upon various community-level properties like stability? Will maximal tolerable overlap be less in the second community? To what extent do the resources available force guild structure? Ecologists are currently seeking answers to such questions. lf Prospects for future empirical work are even brighter, although certainly more difficult and challenging. Well-designed and executed removal and additional experiments or perturbations of equilibrium densities will certainly allow partial quantification of competitive effects, but as Schoener (1974a, 1983) points out, in themselves they probably will not provide much insight into the actual mechanisms of competition. As previously indicated, such experiments will not easily tease apart direct effects from the indirect interactions mediated via other members of ecological communities (Bender et al., 1984). Clever empirical work will probably provide greater insights into competitive mechanisms than further theoretical explorations. In a field manipulation experiment, Dunham (1980) studied competition between two species of rock-dwelling iguanid lizards at Big Bend National Park in Texas using similar, but reciprocal removal experiments. In two dry years, food supplies were apparently scant and removal of the larger lizard species (Sceloporus merriami) had numerous significant effects on the smaller species (Urosaurus ornatus), including increases in density, feeding success, growth rates, lipid levels, and prehibernation body weights. Treatments did not differ from controls during the two wet years, when insect food resources were presumably superabundant. Only one effect of the smaller species on the larger one was evident in these removal experiments: Sceloporus survival was significantly higher in one of the two dry years. Clearly, competition is not reciprocal and varies in intensity from year to year. (To read about ecomorphology, click here) References on Interspecific Competition Case, T. J. 1975. Species number, density compensation, and colonizing ability of lizards on islands in the Gulf of California. Ecology 56: 3-18. Case, T. J. 1979. Character displacement and coevolution in some Cnemidophorus lizards. Fortschr. ZooI. 25: 235-282. Case, T. J. 1983. Niche overlap and the assembly of island lizard communities. Oikos 41: 427-433. Case, T. J. 1983. Sympatry and size similarity in Cnemidophorus. Chapter 14 in R. B. Huey, E. R. Pianka, and T. W. Schoener, (eds.), Lizard ecology: Studies of a model organism (pp. 297-325). Harvard Univ. Press, Cambridge, Mass. Dunhum, A. E. 1980. An experimental study of interspecific competition between the iguanid lizards Sceloporus merriami and Urosaurus ornatus. Ecological Monographs 50: 309-330. Dunham, A. E. 1983. Realized niche overlap, resource abundance, and intensity of interspecific competition. Chapter 12 in R. B. Huey, E. R. Pianka, and T. W. Schoener, (eds.), Lizard ecology: Studies of a model organism (pp. 261-280). Harvard Univ. Press, Cambridge, Mass. Grant, P. R. 1972. Convergent and divergent character displacement. Biological J. Linnaean Society 4: 39-68. Hairston, N.G. 1951. Interspecies competition and its probable influence upon the vertical distribution of Appalachian salamanders of the genus Plethodon. Ecology 32: 266-274. Huey, R. B, E. R. Pianka, M. E. Egan, and L. W. Coons. 1974. Ecological shifts in sympatry: Kalahari fossorial lizards (Typhlosaurus). Ecology 55:304-316. Huey, R. B. and E. R. Pianka. 1974. Ecological character displacement in a lizard. American Zoologist 14: 1127-1136. Jaeger, R. G. 1971. Competitive exclusion as a factor influencing the distribution of two species of terrestrial salamanders. Ecology 52: 632-637. Jaeger, R. G. 1972. Food as a limited resource in competition between two species of terrestrial salamanders. Ecology 53: 535-546. Milstead, W. W. 1961. Competitive relations in lizard populations. In Blair, W. F. (ed.) Vertebrate Speciation. Univ. Texas Press, Austin. Mushinsky, H. R. and J. J. Hebrard. 1977. Food partitioning by five species of water snakes in Louisiana. Her petologica 33: 162-166. Pianka, E. R., R. B. Huey, and L. R. Lawlor. 1979. Niche segregation in desert lizards. Chapter 4 (pp. 67 -115) in Analysis of Ecological Systems, D. J. Horn, R. Mitchell, and G. R. Stairs, eds. Ohio State University Press, Columbus. Rand, A. S. 1964. Ecological distribution in anoline lizards of Puerto Rico. Ecology 45: 745-752. Rand, A. S. 1969. Competitive exclusion among anoles (Sauria: Iguanidae) on small islands in the West Indies. Breviora Mus. Comp. Zool. 327: 1-19. Salzburg, M. A. 1984. Anolis sagrei and Anolis cristatellus in southern Florida: a case study in interspecific competition. Ecology 65: 14-19. Schoener, T. W. 1968. The Anolis lizards of Bimini: resource partitioning in a complex fauna. Ecology 49: 704-726. Schoener, T. W. 1974. Resource partitioning in ecological communities. Science 185: 27-39. Schoener, T. W. 1975a. Competition and the form of habitat shift. Theoret. Pop. Biol. 5: 265-307. Schoener, T. W. 1975b. Presence and absence of habitat shift in some widespread lizard species. Ecol. Monogr. 45: 232-258. Schoener, T. W. 1977. Competition and the niche. Chapter 2 (pp. 35-136) in Biology of the Reptilia, Volume 7, D. W. Tinkle and C. Gans, eds. Academic Press, New York. Schoener, T. W. 1982. The controversy over interspecific competition. American Scientist 70: 586-595. Schoener, T. W. 1983. Field experiments on interspecific competition. American Naturalist 122: 240-285. Schoener, T. W., R. B. Huey, and E. R. Pianka. 1979. A biogeographic extension of the compression hypothesis: Competitors in narrow sympatry. American Naturalist 113: 295-298. Wilbur, H. M. 1972. Competition, predation, and the structure of the Ambystoma-Rana sylvatica community. Ecology 53: 3-21. Wilbur, H.M. and J.P. Collins. 1973. Ecological aspects of amphibian metamorphosis. Science 182: 1305-1314. Back to Herpetology Home Page |